HIPAA Compliant
•
SOC 2 Type II Certified
•
ISO 9001 Certified
•
ISO 27001 Certified
•
GDPR Compliant
Client : Specialty Pharmaceutical Company, United States
Industry : Pharmaceutical — Post-Marketing Pharmacovigilance
Company Size : Mid-Sized
Engagement Duration : Ongoing
Services Used : ICSR Medical Record Review, Pharmacovigilance Narrative Writing
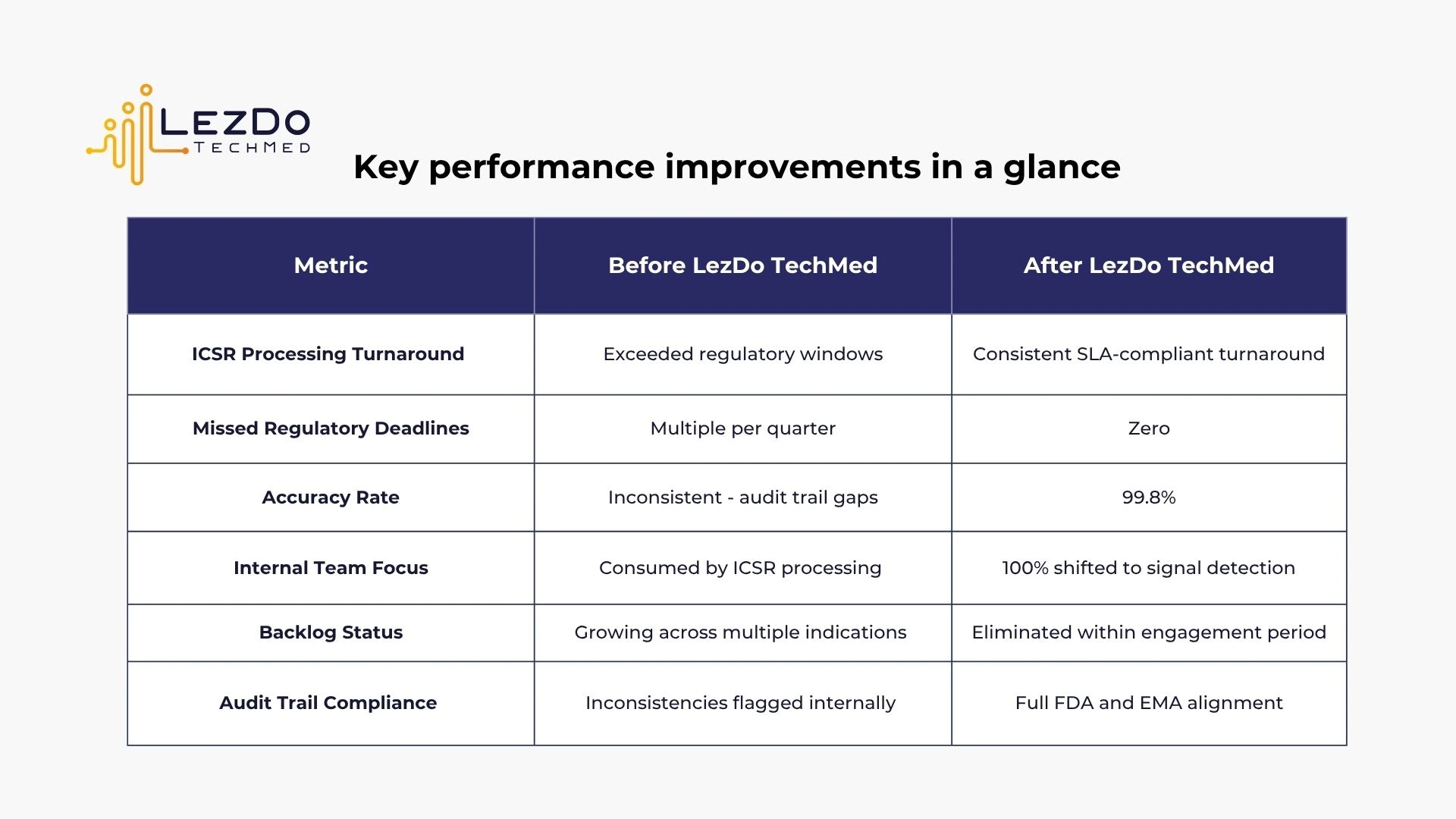
The client lagged behind on processing Individual Case Safety Reports (ICSRs). This delay increased the risk of regulatory issues and kept their internal medical reviewers occupied, even though they were needed for more important tasks like signal detection and updating Risk Management Plans.
Key challenges the client faced:
LezDo TechMed took over ICSR medical record review and narrative writing through CaseDrive, processing every case within consistent SLA timelines with full audit trail documentation aligned to FDA and EMA requirements.
ICSR Medical Record Review
Our medico-legal team reviewed all source documents for each ICSR, including patient records, physician notes, lab results, and adverse event reports. They extracted the clinically relevant information needed for narrative preparation and MedDRA coding support.
Each narrative was structured to meet ICH E2B(R3) requirements and prepared in submission-ready format. The report captured:
All case files were uploaded, tracked, and delivered through CaseDrive, giving the client's PV team real-time visibility into case status, delivery timelines, and audit-ready documentation at every stage of medical data analysis.
LezDo TechMed scaled immediately to absorb the existing backlog across all product indications, without the client needing to hire permanent staff or restructure their internal safety team.
LezDo TechMed integrated directly into the client's existing pharmacovigilance workflow within the first week of engagement. We operated as a dedicated extension of their internal PV team rather than an outsourcing vendor.
The client's case intake process, submission templates, and internal documentation standards were reviewed and mapped into CaseDrive. All incoming ICSRs were routed directly toLezDo TechMed's pharmacovigilance team through a secure, HIPAA-compliant data transfer protocol. Access controls, audit trail parameters, and delivery SLAs were configured and confirmed before the first live case was processed.
While handling live cases, LezDo TechMed dedicated a team to handle the outstanding ICSRs from previous periods. The backlog was eliminated within the first engagement without disrupting the live case processing timelines.
As case volumes fluctuates across product indications, LezDo TechMed scales the processing team up and down without requiring advance notice or contract amendments. Our team absorbs volume spikes that would previously have overwhelmed the internal team.
The client submitted every expedited ICSR within the required 15-calendar-day window —with no Warning Letters, no regulatory queries, and no submission extensions required.
Every ICSR narrative went through a dual-layer review, AI-assisted extraction followed by expert medico-legal quality audit- ensuring clinical accuracy, MedDRA coding consistency, and ICH E2B(R3) compliance before delivery.
Regardless of weekly volume fluctuations, LezDo TechMed consistently delivered within consistent turnaround. This gave the client's PV team the buffer needed to review, approve, and submit on time.
With ICSR processing fully handled, the client's medical reviewer team redirected their full capacity to core tasks, such as signal detection, aggregate safety reporting, and RMP updates.
“LezDo TechMed provided a reliable ICSR processing solution that helped our team meet deadlines consistently. The quality of the narratives reduced rework, allowing our team to focus more on review and signal detection.”
— Clinical Data Operations Lead, US Specialty Pharmaceutical Company
For specialty pharma companies managing post-marketing PV obligations, missed ICSR deadlines are not just an operational problem. They can pose a great regulatory risk withdirect consequences for clinical programs and product approvals.
By outsourcing ICSR processing to LezDo TechMed, this client achieved full regulatory compliance across their entire product portfolio, without adding headcount, restructuring their team, or compromising signal detection work.
A single FDA Warning Letter for late ICSR reporting can trigger enhanced scrutiny across anentire development program. Zero missed deadlines are not just improved efficiency, but a program protection outcome.
With LezDo TechMed’s support, the client transformed its ICSR operations from a backlog-driven process into a streamlined workflow. The result was a complete shift toward proactive pharmacovigilance and better patient safety outcomes.
Ready to eliminate missed PV deadlines? LezDo TechMed’s pharmacovigilance team delivers audit ready ICSR processing within SLA turnaround time, scaling with your program’s requirements.
Anjana Devi Vijay is a Medical–Legal Research Analyst with seven years of experience translating complex medical and legal information into clear, practical insights. Skilled in research, analytics, and deposition summary review, she understands the documentation and workflow challenges faced in the medical–legal field. She creates concise, solution-focused content-including blogs, eBooks, and case studies- that helps attorneys, evaluators, and claims professionals improve decision-making and strengthen case outcomes.