HIPAA Compliant
•
SOC 2 Type II Certified
•
ISO 9001 Certified
•
ISO 27001 Certified
•
GDPR Compliant
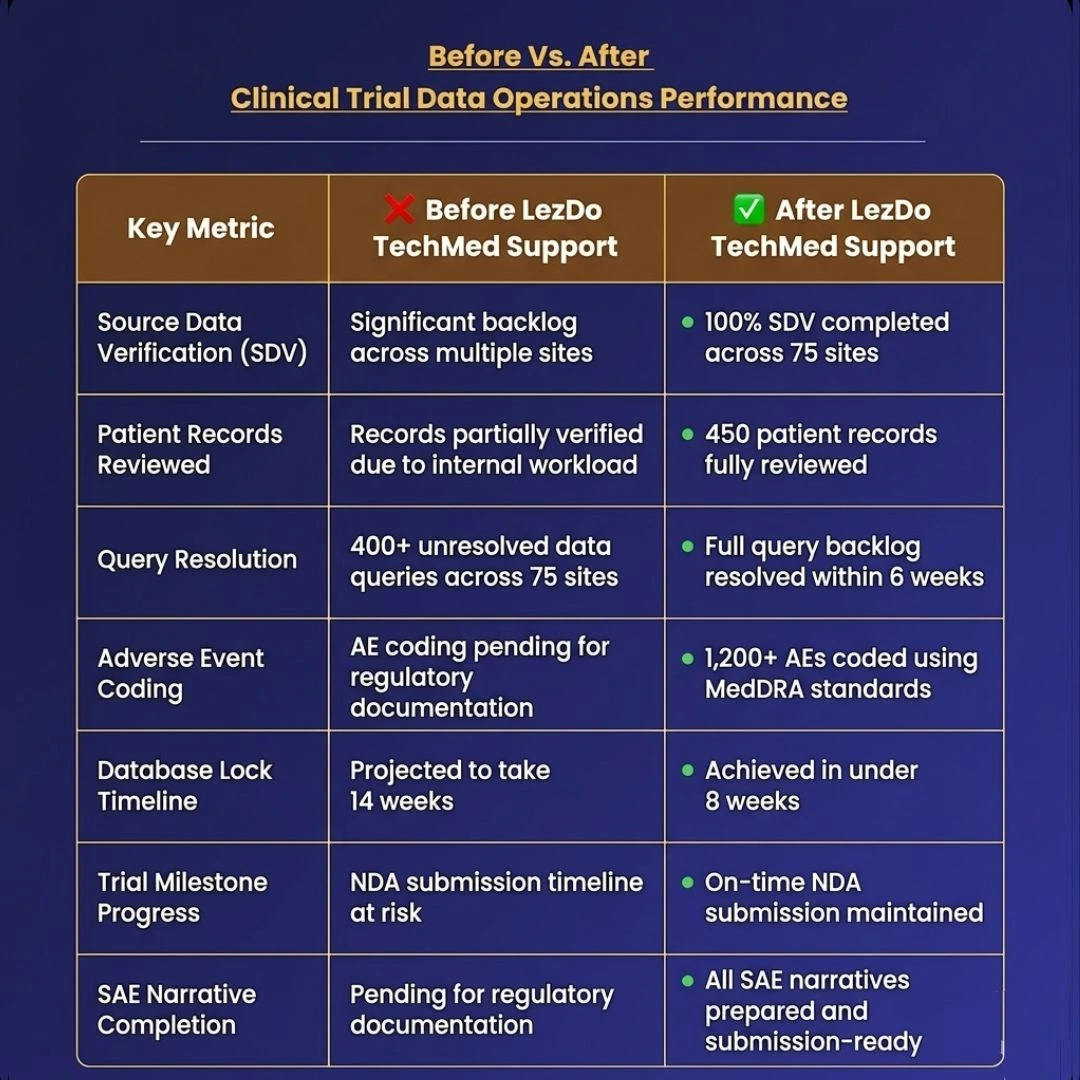
We worked with a New York-based biotech company that focuses on cancer research. They were in the stretch of a massive global study involving 450 patients across 75 different clinics. They had reached a huge "make or break" moment: it was finally time to bundle up all their research and submit it to the government to get their new cancer drug approved for the market.
As the trial moved into its final phase of data collection, challenges began to increase. There was a rise in query volumes, unresolved data issues, and a growing backlog in Source Data Verification (SDV). These factors started putting pressure on the internal CRO team.
With the database lock timeline approaching, the risk of delays became a serious concern. Any slowdown at this stage could affect the NDA submission timeline and delay the sponsor’s regulatory plans and time to market.
To manage these challenges and keep the trial on track, the sponsor partnered with LezDo TechMed for scalable clinical data support.
Large Phase III oncology trials generate complex clinical datasets across multiple sites. As the study progressed, several operational bottlenecks emerged:
For a Phase IIIoncology program of this scale, even a small delay can have major financial consequences. A delayed NDA submission can cost sponsors over $1 million per month in lost market access opportunity.
The company needed a partner who could move fast and grow with the project without ever dropping the ball on quality or strict medical rules.
LezDo TechMed brought in a specialized team of data experts to get everything back on track. Our mission was simple: speed up the paperwork and double-check every single detail. We didn't cut corners, we just worked smarter. By following the "gold standard" of clinical rules (like GCP and 21 CFR Part 11), we ensured the company stayed fully compliant and ready for regulation.
We made sure every step followed the "gold standard" of medical rules (like GCP and 21CFR Part 11), keeping the company fully compliant with government regulations.
Our team performed a "100% match" check. They compared every single physical patient record against the digital database to make sure they were identical. By catching and fixing these tiny mismatches early, we made the study’s data much more reliable and ready for the finish line.
Clinical documentation specialists extracted relevant patient history data from medical records and structured it for database alignment, improving accessibility for investigators and study managers.
LezDo TechMed’s team worked directly with CRO data managers and site coordinators to review documentation, validate discrepancies, and resolve outstanding queries, clearing the backlog and improving data readiness.
We coded more than 1,200 adverse events using MedDRA standards, making sure every medical issue was labeled correctly and ready for official review.
The team also prepared Serious Adverse Event (SAE) narratives required for regulatory documentation, supporting safety reporting and submission readiness.
The engagement was executed over 11 weeks, from initial onboarding through database lock support.
The execution model focused on structured workflows and strict quality assurance:
This structured operational framework allowed the team to rapidly clear the backlog while maintaining regulatory-grade data quality.
Comprehensive clinical documentation review ensured data accuracy across all participating sites.
Standardized safety coding improved clarity and consistency of adverse event reporting.
What was supposed to take 14 weeks was finished in under 8, getting the study to the finish line over a month ahead of schedule.
Most importantly, the sponsor preserved the regulatory submission timeline and avoided a potential delay in market access.
For late-stage clinical trials, operational delays can have significant downstream consequences.
By maintaining the planned submission timeline, the sponsor avoided a potential 6–8 week regulatory delay, which could have resulted in millions of dollars in lost revenue opportunity due to delayed market entry.
By restoring clinical data operations to full capacity within 11 weeks, LezDo TechMed enabled the sponsor to achieve database lock ahead of the original 14-week projection and proceed with NDA submission on schedule. For a Phase III oncology program approaching commercialization, that outcome represented not just a preserved timeline — but a preserved market opportunity.
"LezDo TechMed's team integrated into our trial operations within days. Their ability to handle the SDV backlog while simultaneously managing AE coding gave us the confidence to proceed with NDA submission on schedule."
— Clinical Operations Director (Client identity withheld under confidentiality agreement)
Reviewed By:
A Senior Clinical Data Manager with 6+ years of Phase II–III trial support experience
LezDo TechMed Clinical Operations Team.
Anjana Devi Vijay is a Medical–Legal Research Analyst with seven years of experience translating complex medical and legal information into clear, practical insights. Skilled in research, analytics, and deposition summary review, she understands the documentation and workflow challenges faced in the medical–legal field. She creates concise, solution-focused content-including blogs, eBooks, and case studies- that helps attorneys, evaluators, and claims professionals improve decision-making and strengthen case outcomes.